Why is teflon so slippery?
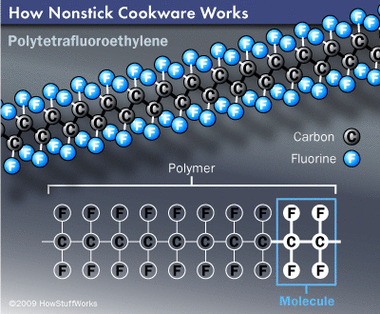
In order to understand why polytetrafluoroethylene (PTFE) is a useful nonstick coating, we must take a closer look at its structure and properties. PTFE is a type of fluoropolymer. A polymer is a large molecule made up of smaller molecules of the same type. Fluoropolymers are polymers that include the fluorine atom, which is the key to many of the unique characteristics of PTFE. According to William Raiford, technology manager with DuPont Fluoroproducts, fluorine's electron structure is very stable -- it doesn't share its electrons with other atoms easily.
The chemical composition of PTFE is based on two carbon atoms and four fluorine atoms, which are repeated throughout the molecule's structure. "The molecular structure has an outer shell filled with fluorine atoms," says Raiford. "The fluorine atoms shield the carbon atoms from reacting."
Think about a high school dance. The fluorine atoms are the chaperones, and the carbon atoms are the female students. Anything that might interact with the carbon is the male students. As chaperones, the fluorine atoms keep the boys from interacting with the girls -- in a frying pan, it keeps the food from sticking.
Along with their resistance to reacting with other chemicals, fluorine atoms also play a role in giving PTFE a low coefficient of friction. The coefficient of friction is a measurement of how easy it is for two substances to slide by each other -- the harder it is to slide, the higher the coefficient of friction. PTFE doesn't put up a lot of resistance when sliding by another substance, giving it a low coefficient of friction. The low coefficient of friction gives PTFE that signature slippery quality.
PTFE is not the only fluoropolymer used to make nonstick cookware. According to Raiford, other polymers of the same family have been developed with slightly refined structures to offer easier application, more strength or decorative looks to the nonstick surface.
The chemical composition of PTFE is based on two carbon atoms and four fluorine atoms, which are repeated throughout the molecule's structure. "The molecular structure has an outer shell filled with fluorine atoms," says Raiford. "The fluorine atoms shield the carbon atoms from reacting."
Think about a high school dance. The fluorine atoms are the chaperones, and the carbon atoms are the female students. Anything that might interact with the carbon is the male students. As chaperones, the fluorine atoms keep the boys from interacting with the girls -- in a frying pan, it keeps the food from sticking.
Along with their resistance to reacting with other chemicals, fluorine atoms also play a role in giving PTFE a low coefficient of friction. The coefficient of friction is a measurement of how easy it is for two substances to slide by each other -- the harder it is to slide, the higher the coefficient of friction. PTFE doesn't put up a lot of resistance when sliding by another substance, giving it a low coefficient of friction. The low coefficient of friction gives PTFE that signature slippery quality.
PTFE is not the only fluoropolymer used to make nonstick cookware. According to Raiford, other polymers of the same family have been developed with slightly refined structures to offer easier application, more strength or decorative looks to the nonstick surface.
Sticking it to the pan
Polytetrafluoroethylene (PTFE) has a unique slickness and inactivity with other chemicals that make it very useful in many applications -- it's a component in space suits, and it insulates high-voltage wires. Yet these characteristics also pose challenges when it comes to creating nonstick cookware. The nonstick coating must somehow stick to the surface of the pan.
There are many variations of the process to meld the nonstick coating onto the cookware's metal surface. Basically, they all start with the metal base, or substrate, which is created in the shape of the desired cookware. Most nonstick cookware is made of aluminium, but other metals, such as stainless steel, are also used.
The next part of the process deals with applying the nonstick coating to the pan substrate. According to DuPont technology manager William Raiford, there are two ways to overcome the nonstick problem -- mechanically and chemically.
Let's go back to the example of the dance. The chaperones keep the female students from interacting with the male students. To distract the chaperone, a male student might enlist a friend to create a disturbance on the other side of the room, causing all the chaperones to move in that direction. Another tactic might be to build some common ground among the chaperones and students by playing some music from the chaperones' era and getting everyone onto the dance floor together. Either way, the male dancers have a chance to dance with the females.
When making nonstick cookware, the manufacturing version of the male students' disturbance starts with roughening the surface of the substrate. This makes it easier for PTFE's fluorine molecules to stick to the surface. Some methods include roughing the surface with molten metal or chemicals.
The common ground is a primer or base coat, which has a special formulation that allows it to adhere to both the metal substrate and several nonstick coating layers. The number of PTFE-based coatings, which are either sprayed or rolled onto the surface, depends on the type of pan.
According to Raiford, this process can include heating between each layer, or the layers can be put on top of each other while they are still wet. The final step is sintering. "This is a high temperature bake usually at around 800 degrees Fahrenheit for about three to five minutes," Raiford says -- that's about 427 degrees Celsius. "This dries and cures the polymer and also helps to lock it to the metal."
There are many variations of the process to meld the nonstick coating onto the cookware's metal surface. Basically, they all start with the metal base, or substrate, which is created in the shape of the desired cookware. Most nonstick cookware is made of aluminium, but other metals, such as stainless steel, are also used.
The next part of the process deals with applying the nonstick coating to the pan substrate. According to DuPont technology manager William Raiford, there are two ways to overcome the nonstick problem -- mechanically and chemically.
Let's go back to the example of the dance. The chaperones keep the female students from interacting with the male students. To distract the chaperone, a male student might enlist a friend to create a disturbance on the other side of the room, causing all the chaperones to move in that direction. Another tactic might be to build some common ground among the chaperones and students by playing some music from the chaperones' era and getting everyone onto the dance floor together. Either way, the male dancers have a chance to dance with the females.
When making nonstick cookware, the manufacturing version of the male students' disturbance starts with roughening the surface of the substrate. This makes it easier for PTFE's fluorine molecules to stick to the surface. Some methods include roughing the surface with molten metal or chemicals.
The common ground is a primer or base coat, which has a special formulation that allows it to adhere to both the metal substrate and several nonstick coating layers. The number of PTFE-based coatings, which are either sprayed or rolled onto the surface, depends on the type of pan.
According to Raiford, this process can include heating between each layer, or the layers can be put on top of each other while they are still wet. The final step is sintering. "This is a high temperature bake usually at around 800 degrees Fahrenheit for about three to five minutes," Raiford says -- that's about 427 degrees Celsius. "This dries and cures the polymer and also helps to lock it to the metal."
